Press Release
Durable Wood Carbon Sponge Could Be the Future of Wearable Sensors, Pollutant Treatment
FOR IMMEDIATE RELEASE March 1, 2018
CONTACT:
Natifia Mullings
301-405-4076
mullings@umd.edu
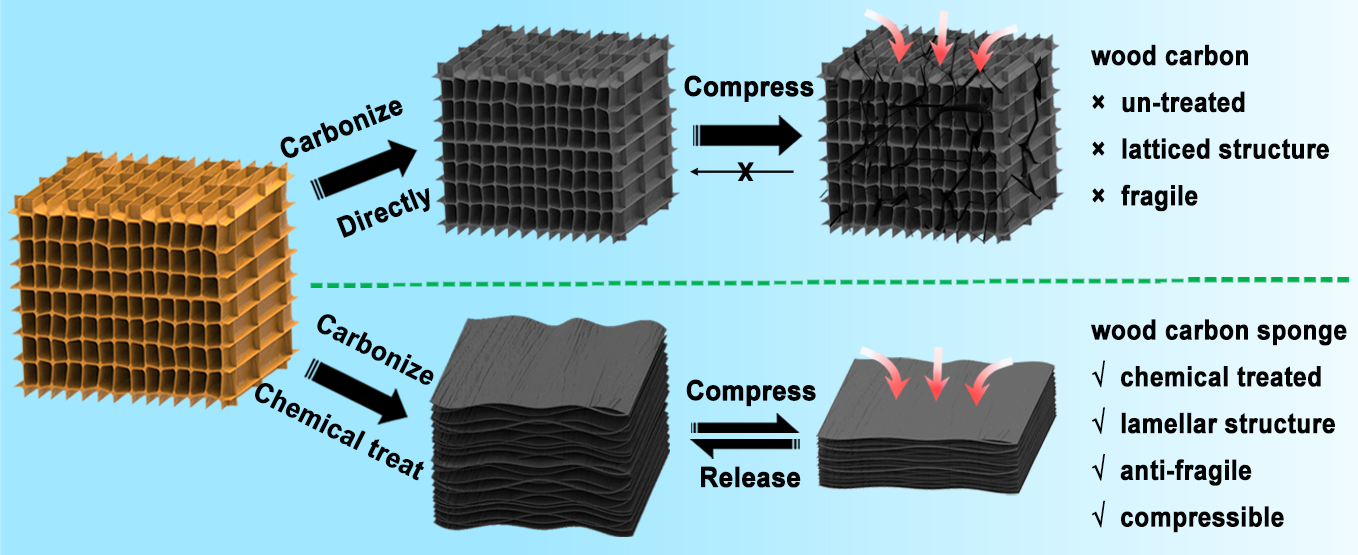
Rigid wood can be directly transformed into a compressible wood carbon sponge by a simple chemical treatment and carbonization.
College Park, Md. — Engineers at the University of Maryland, College Park (UMD) have for the first time demonstrated that wood can be directly converted into a carbon sponge capable of enduring repeated compression and other extreme mechanical conditions. The UMD engineers’ wood carbon sponge overcomes several limiting factors of other lightweight, compressible carbon sponges because it is simpler, less expensive, and more sustainable to produce. The new sponge can be used in various applications such as energy storage (e.g., batteries), pollutant treatment, and electronic devices and sensors.
A peer-reviewed paper about the research was published March 1 in the journal Chem.
Most lightweight, compressible carbon sponges are made from raw materials that are usually unrenewable fossil resources—such as graphene—and by a complicated fabrication process that involves multiple steps and environmentally unfriendly chemicals. In contrast, the UMD researchers use a simple chemical process to transform balsa wood, a choice biomass-based material that is both renewable and abundant.
“Our results reveal that rigid and incompressible balsa can be made highly compressible by a chemical treatment and carbonization process, yielding a wood carbon sponge with mechanical compressibility and fatigue resistance and electrical response sensitivity surpassing those of most reported compressible carbonaceous materials,” says corresponding author Liangbing Hu, associate professor of materials science and engineering at UMD’s A. James Clark School of Engineering.
Hu and colleagues achieved the bendable yet resilient architecture of the wood carbon sponge by using common chemicals to destroy the stiff hemicellulose and lignin fibers that maintain the normal cell-wall structure of balsa wood, then heating the treated wood to 1,000C in order to turn the organic material into carbon alone. The net effect of the process was to collapse the repeated, regular, rectangular pockets typical of the microstructure of balsa and other woods and replace them with a stack of wavy, interlocking, arch-like carbon sheets, likened by Hu to a cross between a coiled spring and a honeycomb.
Normal carbonized wood—obtained from only the heating step without any chemical modifications—is so fragile that any reasonable applied force pulverizes it irreversibly into ash and dust. However, the wood carbon sponge withstood and rebounded from substantial compression for up to 10,000 consecutive trials before deformation set in. Such a performance initially surprised the research team.
“Our process for creating the wood carbon sponge is unique because we preserve the structure of the wood. This makes the sponge highly compressible and resistant to stress. This means the performance of our wood carbon sponge is one of the best among all lightweight and compressible carbonaceous materials ever reported,” says lead author Chaoji Chen, postdoctoral researcher at UMD’s A. James Clark School of Engineering.
After conducting further mechanical and electrical tests on the sponge, the researchers were able to incorporate a slice of it into a strain sensor prototype suitable for attachment to a human finger, a quality desirable for use in wearable fitness or health-monitoring electronics.
The researchers believe that the wood carbon sponge could also be incorporated into water purification devices and energy storage and conversion technologies, such as supercapacitors and rechargeable batteries. “The abundant applications illustrate the value of a strategy that explores the hidden potentials of natural materials, such as trees, by drawing inspiration from other natural structures and sources,” Hu says.
More Information:
This work was supported by the Maryland NanoCenter.
Chem, Chen et al.: “Scalable and sustainable approach toward highly compressible, anisotropic, lamellar carbon sponge.” DOI: 10.1016/j.chempr.2017.12.028
The A. James Clark School of Engineering at the University of Maryland serves as the catalyst for high-quality research, innovation, and learning, delivering on a promise that all graduates will leave ready to impact the Grand Challenges (energy, environment, security, and human health) of the 21st century. The Clark School is dedicated to leading and transforming the engineering discipline and profession, to accelerating entrepreneurship, and to transforming research and learning activities into new innovations that benefit millions. Visit us online at www.eng.umd.edu and follow us on Twitter @ClarkSchool.